
Dimagnesium Phosphate
Also known as Magnesium hydrogen phosphate and Magnesium phosphate dibasic
What is Dimagnesium phosphate?
Dimagnesium phosphate (DMP) is a food additive used in baked goods as a leavening agent and nutritional supplement, specially in infant formulations.1
As an acidulant in baking powders, it has a neutralizing value of 40 and may provide metallic off-flavors due to an excess of phosphate content.1
- Dimagnesium phosphate is found as wihte crystalline powder.
- It is obtained from a phosphoric acid reaction with magnesium oxide.2
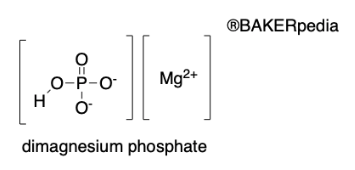
Chemical Formulation
Origin
Dimagnesium phosphate is produced from the reaction of phosphoric acid and magnesium oxide or magnesium hydroxide.2
Phosphoric acid is produced by two routes:
- Wet process that involves the digestion of phosphate rocks with a mineral acid
- Electrothermal process.2
Function
Dimagnesium phosphate main functions in baked goods include:1
- Leavening: typically used in combination with sodium bicarbonate to provide leavening for baked goods that is heat-activated only during baking.
- Nutritional supplement: as a fortificant and good source of phosphates and magnesium in infant formulas.
Nutrition
Dimagnesium phosphate is used as a food additive and thus provides no calories. As a nutritional supplement, DMP is used to improve phosphate content of infant formulas.2
The current established average daily intake (ADI) of phosphates expressed as phosphorus is 40 mg/kg per day, which corresponds to 2800 mg/day of phosphorus for a 70 kg person.2
Commercial production
Dimagnesium phosphate is commercially produced through the following process:2
- Raw materials procurement and weighing: adequate proportions of phosphoric acid and magnesium oxide are procured and weighed.
- Mixing: phosphoric acid and magnesium oxide are mixed in a reaction vessel to produce magnesium phosphate.
- Centrifugation: magnesium phosphate is separated from the reaction mixture by centrifugation or by filtration.
- Drying: magnesium phosphate is dried to the appropriate moisture content.
- Milling: the product is milled to the desired particle size using a series of sieves.
- Metal detection: the salt is passed through a metal detector to remove potential contaminants before packaging.
Application
Dimagnesium phosphate is commonly used in the manufacture of:1
- Cakes and cakes mixes
- Doughnuts
- Muffins
- Waffles
- Wafers
- Pancakes
- Refrigerated doughs
- Baking powders
- Nutritional supplements
DMP is a heat activated leavening agent at 40.5 – 43.5 oC (104.9 – 110.3 oF), an intermediate value between sodium aluminium phosphate (SALP) and dicalcium phosphate dihydrate (DCP-2). As a slow acting leavening agent, the use of this salt in combination with a faster leavening agent is recommended.
It has a dough reaction rate of 24 after heat activation.2,3
Dimagnesium salt provides a neutral flavor and is characterized by being the first sodium free leavening agent acidulant.3
Regulations
Dimagnesium phosphate is considered GRAS by the FDA when used following good manufacturing practices.4
In the EU, dimagnesium phosphate (E 343 ii) is considered safe by the EU Commission Regulation No 1333/2008 Annex II and III. However, some concerns have been raised in Europe regarding current daily intake of phosphate of infant foods.2
References
- Branen, A. L, Davidson, P.M, Salminen, S and Thorngate, J.H. Food additives. CRC Press, 2001.
- Younes, M., et al. “EFSA Panel on Food Additives Flavourings, Re-evaluation of phosphoric acid–phosphates–di-, tri-and polyphosphates (E 338–341, E 343, E 450–452) as food additives and the safety of proposed extension of use.” EFSA Journal 17.6 (2019): 5674.
- Tiefenbacher, Karl F. The Technology of Wafers and Waffles I: Operational Aspects. Academic Press, 2017.
- Food and Drug Administration (FDA). US Department of Health and Human Services. CFR Code of Federal Regulations Title 21, Part 184 Direct Food Substances Affirmed As Generally Recognized As Safe, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=184.1434 , Accessed 23 October 2021.

