Vanillin
Also known as Vanilla imitation
What is Vanillin?
Vanillin is the predominant compound in vanilla that imparts pleasant aromatic flavor notes. It is an aldehyde that can replace vanilla extract, and is referred to as vanilla imitation. As the world’s most popular flavor, vanillin is used in beverages and food products such as ice cream, dairy products, chocolates, cakes and other baked products.1
- Extraction of natural vanillin from vanilla beans is labor intensive, thus its use in food products is cost prohibitive.
- Therefore, about 90% of the world’s vanillin is synthetically obtained from guaiacol, lignin or other substrates .1
Origin
Vanilla (Vanilla planifolia) is thought to have originated in Central and North America, mainly Mexico. Now, the Comores, Madagascar and Reunion are the main producers of vanilla. Various types of vanilla beans such as Bourbon, Tahitian and Indonesian are known with various flavor profiles.1 Today, vanillin is often produced synthetically from guaiacol or other substrates.
Chemical formula
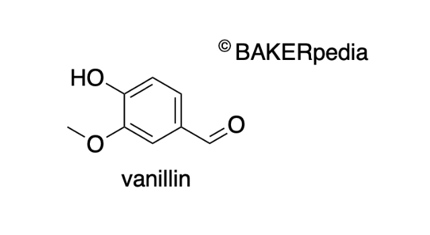
4-hydroxy-3-methoxybenzaldehyde
Function
Vanillin can serve several purposes in baked goods:1
- Flavor and aroma: provides a sweet taste and a rich vanilla flavor and aroma.
- Antimicrobial effects: may serve as food preservative in several dairy products.
Nutrition
Typical nutritional value of commercially available vanillin or “vanilla imitation”per 100 g:2
| Component | Grams |
|---|---|
| Water | 64.5 |
| Carbohydrate | 2.41 |
| Ash | 0.22 |
| Protein | 0.05 |
*Composition made not add to a 100 g
Vanillin has a demonstrated antioxidant, antimutagenic, anticlastogenic and anticarcinogenic activities.1
Commercial production
Main processes for commercial production of vanillin include:
Biological synthesis from vanilla bean 1
- Procurement of matured vanilla beans (9 months).
- Scalding: green vanilla beans are placed in hot water at 60 – 70 oC (140 – 158 oF) for 1 – 2 min.
- Autoclaving: vanilla beans from the previous step are later stored in boxes for 2 – 5 days.
- Sunning and sweating: vanilla beans are spread on blankets and placed in the sun and collected at night. This step is repeated for two weeks.
- Drying: for another 2 – 4 weeks or longer until they reach a water content of 25 – 40%.
- Storage for flavor development: beans are stored for several months in small bundles in sealed boxes.
Chemical synthesis from guaiacol 3
- Condensation of guaiacol with glyoxylic acid at room temperature in weak alkaline medium.
- Air oxidation in the presence of a catalyst..
- Acidification and decarboxylation to obtain crude vanilla after hydrolysis of vanillin glucoside.1
- Vacuum distillation
- Recrystallization
Application
Vanillin is used in several baked goods as a primary or secondary flavor, when using vanilla or vanillin in baked goods some consideration should be taken into account:4
- Product composition and baking temperature:
- Low fat content, higher vanilla usage level.
- High protein content, higher vanilla usage level.
- High temperature, higher vanilla usage level.
- Spray dried or encapsulated vanilla flavor is preferred to liquids.
- When using liquid vanilla, mix with the fat and add in the late stages of mixing.
- Low temperatures help preserve the vanilla during baking or cooking.
Regulations
Vanillin is considered GRAS by the FDA, and it has standardized requirements of a maximum ethyl alcohol content of 35%.5
In the EU, vanillin is considered safe; however, EU regulations distinguish between natural and synthetic vanillin. The use of catalyst or other enzymes is not allowed in the production of natural flavors.5
References
- Berger,R. G. Flavours and Fragrances: Chemistry, Bioprocessing and Sustainability. Alemania, Physica-Verlag, 2007, pp 203 – 215.
- U.S. Department of Agriculture, Agricultural Research Service. FoodData Central, 30 March 2021.https://fdc.nal.usda.gov/fdc-app.html#/food-details/1506684/nutrients . Accessed 24 April 2021.
- Bauer, K, Garbe,D and Surburg,H. Common Fragrance and Flavor Materials: Preparation, Properties and Uses. John Wiley & Sons, 2008, 290 pp .
- Havkin-Frenkel,D and Belanger, F.C. Handbook of Vanilla Science and Technology. United Kingdom: Wiley, 2018.
- Grocholl, L. “Vanilla Regulations”. Sigmaaldrich.Com, 2021, https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/food-and-beverage-testing-and-manufacturing/flavor-and-fragrance-formulation/vanilla-regulations . Accessed 29 June 2021.


