
Alpha-cyclodextrin
Also known as alpha-dextrin (α-CD) or alpha-cycloamylose
What is Alpha-cyclodextrin?
Alpha-cyclodextrin has emulsifying and stabilizing properties. It can also eliminate undesirable compounds. In the food industry, alpha dextrins are used for:
- Stabilizing oil-in-water emulsions
- Controlling the solubility and dispersibility of un-mixable liquids
- Masking odors and unwanted flavors
- Stabilizing and protecting compounds from light, heat and oxidation
- Entrapment of cholesterol
Origin
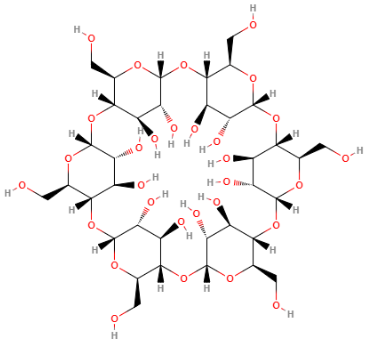
Alpha dextrin is made from starch, using enzymatic conversion. Furthermore, it is water-soluble, sugar ring molecule. It is a non-reducing cyclic saccharide originated from starch. Chemically, it consists of 6 alpha-1,4-linked glucose units. Together, they form a truncated cone or donut structure with a hydrophilic outer surface and a large hydrophobic hole in its center.1
Commercial production
Alpha dextrin is produced from natural plant starch. This is done via an enzymatic conversion process using the food grade enzyme, cyclodextrin glycosyltransferase (glucanotransferase). The enzyme forms rings of 1,4-linked α-D-glucopyranosyl units.2,3
The enzyme can form 6-, 7-, and 8-membered cyclic maltooligosaccharides. It does this by means of an intramolecular transfer (cyclization).2
Function
The main function of alpha-cyclodextrin in food products is the stabilization of emulsions as an alternative to common emulsifiers. It can act as surfactants in food, allowing water-and-fat dispersions to be evenly mixed. This forms stable emulsions.
The presence of hydrophilic groups on the outside of the alpha-cyclodextrin makes the molecule water-soluble. The hydrophobic center can trap nonpolar molecules which fit within the alpha-cyclodextrin’s cavity.1,3
Alpha dextrin has the unique ability of complexing nonpolar substances. It functions as a carrier or stabilizer for fats, essential oils, organic acids, alcohols, flavors, and many others. Such inclusion complexes are termed clathrates, which are chemical substances that traps or contains molecules. The complexed lipophilic molecules are known as guest molecules.1,3
Application
Alpha-cyclodextrin can be used in the production of icings, batter cakes, sponge cakes, mayonnaise, and many more products. They can also be used as an emulsifier alternative in gluten- and egg-free bakery products.
Special considerations for using alpha-cyclodextrin as emulsifier in bakery products:
- Formulations containing amylase: alpha-amylase can hydrolyze α-1,4-glycosidic bonds of alpha-cyclodextrin. Formulations that contain amylase for dough conditioning and crumb softening could impact the function of alpha-cyclodextrin.
- Solubility in water: alpha-cyclodextrin has a solubility of 13 g/100 mL of water at 77°F (25°C). However, their water solubility increases to 66 g/100 mL when the temperature is raised to 140°F (60°C).2
- Formulations rich in acids: a proper balance should be established between the total titratable acidity (TTA) and alpha-cyclodextrin to avoid hydrolysis. As a result, the cyclodextrin’s functionality could be hurt.
- Emulsion preparation: for better results, alpha-cyclodextrin should be first dispersed in water. Then, fat or oil can be gradually added to the mixture to create a stable emulsion. Meanwhile, stirring the mixture to evenly disperse the discontinuous phase and surfactant.
FDA regulation
Alpha dextrin is considered GRAS in accordance with GRAS Notice 155.4
References
- Amiri, S., and Amiri, S. “Cyclodextrin Applications.” Cyclodextrins. Properties and Industrial Applications, John Wiley & Sons Ltd, 2017, pp. 269–288.
- Hedges, A. “Cyclodextrins: Properties and Applications.” Starch: Chemistry and Technology, 3rd edition, Academic Press, Elsevier Inc., 2009, pp. 833–851.
- BeMiller, J.N. “Starches: Conversions, Modifications, and Uses.” Carbohydrate Chemistry for Food Scientists, 3rd edition, Elsevier Inc. / AACC International., 2019, pp. 191–221.
- U.S. Food and Drug Administration. “GRAS Notices.” Dec 20, 2004, https://www.accessdata.fda.gov/scripts/fdcc/?set=GRASNotices&id=155&sort=GRN_No&order=DESC&startrow=1&type=basic&search=155. Accessed 22 September 2019.

