Sodium Iodate
What is Sodium Iodate?
Sodium iodate is the sodium salt of iodic acid. It is an oxidizing agent and dough conditioner used to strengthen doughs. It is commonly used in leavened products such as bread, rolls, and sweet rolls.1 It is commonly found as a white crystalline powder and has the following physical properties:1
- Chemical formula: NaIO3
- Molecular weight: 197.892 g/mol
- White, crystalline powder
- Water and acetic acid soluble
Chemical Structure
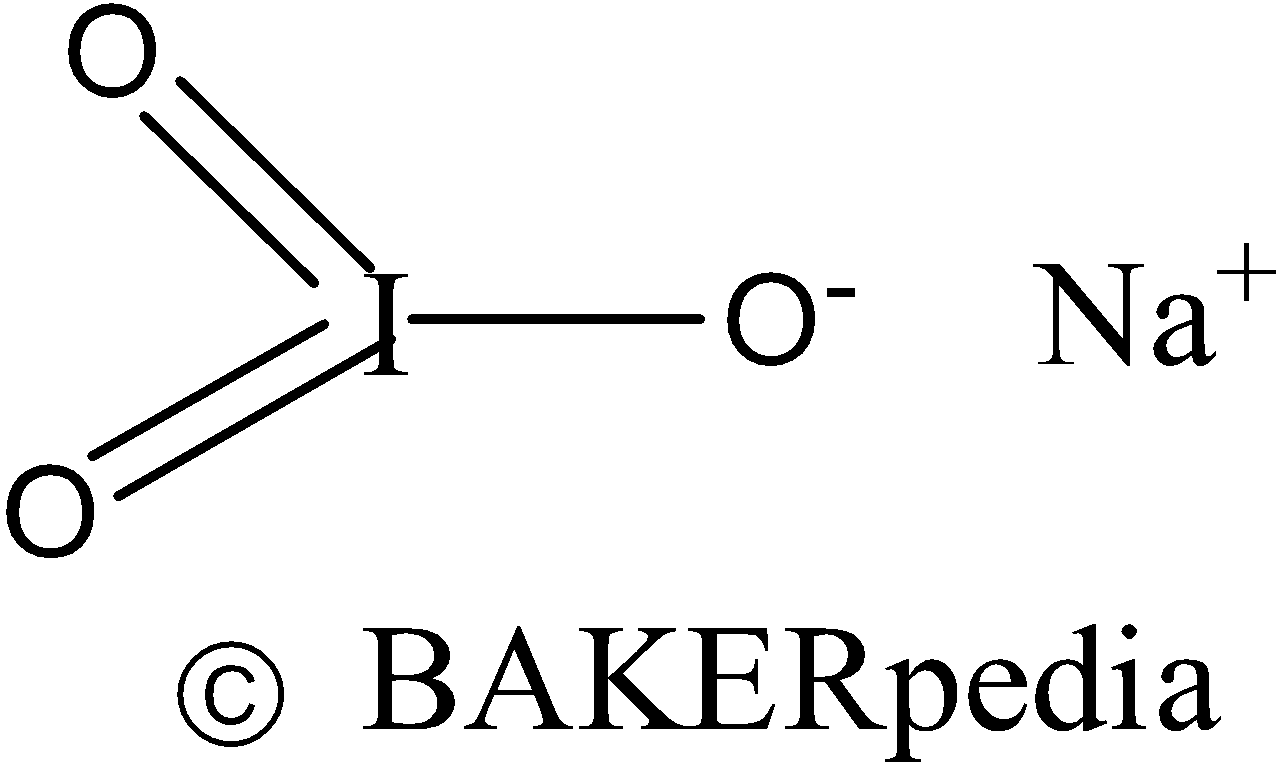
Origin
Iodine, found in plants from marine sources, is the basic raw material for making sodium iodate via its reaction with sodium chlorate in the presence of nitric acid. An alternative method relies on the oxidation of iodine followed by neutralization with a hydroxide.1
Among oxidizing agents, sodium iodate is less commonly used. Instead, potassium iodate and calcium iodate or peroxides are preferred in baking.2
Function
In baked goods, this ingredient performs several functions:2
- Dough conditioner: as an oxidizing agent, it aids in gluten network strengthening via oxidizing the protein’s thiol group.
- Texture: improves loaf volume and crumb texture.
Nutrition and health
Sodium iodate is a source of iodine, an essential nutrient for the synthesis of thyroid hormones (thyroxine and triiodothyronine) involved in controlling basal metabolic rate and regular growth and development.3
A deficiency of this trace element is associated with hypothyroidism, weight gain and enlarged thyroid gland.3
Commercial production
It can be commercially produced by the following processes:1
- Reaction of sodium chlorate and iodine in the presence of nitric acid.
- Oxidation of iodine to iodic acid, and further neutralization with an oxide or a concentrated hydroxide such as sodium hydroxide.
- Oxidation reaction of iodides to iodates.
Application
Sodium iodate is not the most preferred oxidizing agent for dough conditioning. Alternatives such as potassium bromate and ascorbic acid are frequently used partly due to their availability and processing condition adaptability, although it may negatively impact taste.
As an oxidizing agent, sodium iodate is considered a fast oxidizer which requires care when developing formulations and processing methods.2 When used in breadmaking, sodium iodate should be added directly to the flour where it immediately starts reacting with the gluten thiol group.
Usage levels of sodium iodate are not established by the FDA or EU Commission; however common usage levels of similar dough conditioner are shown in the following table:2
| Oxidizing Agent | Usage Level |
|---|---|
| Potassium Iodate | 10 -75 ppm |
| Calcium Iodate | 10 -75 ppm |
Benefits of using oxidizing agents, like sodium iodate:2
- Improves machinability of the dough
- Improves gas retention
- Provides good volume and uniform crumb grain.
The use of sodium iodate, and other oxidizing agents is inadvisable in cookies, cakes and biscuits.
Regulations
Sodium iodate is considered GRAS by the FDA when used as a trace mineral in animal feed.4
In the EU, sodium iodate can be added to food products such as cereal products and as a mineral supplement. Its use in food products is regulated by the EU Commission No 1925/2006 and 609/2013. However, this ingredient doesn’t have an E number, and thus is not fully recognized as a safe food additive.5
References
- PubChem.”Sodium Iodate”. Pubchem.Ncbi.Nlm.Nih.Gov, 2020, https://pubchem.ncbi.nlm.nih.gov/compound/Sodium-iodate . Accessed 25 Dec 2020.
- Lallemand Inc. Lallemand Baking Update. Volume 3. Number 6. Available at: https://www.lallemandbaking.com/wp-content/uploads/2018/04/3_6DOUGH.pdf Accessed 25 Dec 2020.
- Ronzio, R. A. The encyclopedia of nutrition and good health. Infobase Publishing, 2003.
- Food and Drug Administration (FDA). US Department of Health and Human Services. CFR Code of Federal Regulations Title 21, Part 582 Food Substances Generally Recognized As Safe, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=582.80 , Accessed 25 December 2020.
- European Commission (Ec). Commission Regulation Regulation (Eu) No 609/2013on food intended for infants and young children, food for special medical purposes, and total diet replacement for weight control and repealing Council Directive 92/52/EEC, Commission Directives 96/8/EC, 1999/21/EC, 2006/125/EC and 2006/141/EC, Directive 2009/39/EC of the European Parliament and of the Council and Commission Regulations (EC) No 41/2009 and (EC) No 953/2009,. Official Journal Of European Communities, 12 June 2013.


