
Modified Wheat Starch
What is Modified Wheat Starch?
Modified wheat starch is simply wheat starch that has been modified for specific uses. Wheat, corn, and any other cereal starch, can be modified in the following ways:
- Enzymatically
- Physically
- Chemically
Objectives of starch modification include:
- Increase versatility of starch to be used in multiple applications
- Impart enhanced properties compared with their native precursors
- Diminish detrimental characteristics of starch
Origin
Wheat was the most popular and widespread starch for many years, due to the abundance of the grain. As cultivation of potatoes and maize grew during the 18th century, they became common sources of starch as well. As processed and packaged foods gained popularity, the need for creating specific attributes from the carbohydrate emerged. Modification processes developed very quickly and FDA regulations evolved to assess the types of modifications allowed for food use.1
Function
Various starch modification approaches resulted in new starches that can impart some of the following benefits to food products:
- Thickening (viscosity building) of liquid foods at room or cold temperatures
- Superior freeze-thaw stability
- Polymeric structure formation in gluten-free baked goods
- Gelling ability at lower temperatures
- Adhesion
- Superior moisture control
- Shelf-life extension (anti-staling)
- Encapsulation of volatile aromas and flavors added to food products
Modified wheat starch can be used in infinite products, such as:
- Batters
- Yeast-leavened doughs
- Breakfast cereals
- Toppings
- Soups
- Sauces
- Pudding-type products
- Icings
- Pie fillings
- Whipped cream
- Jellies
- Meringues
- Foam-type cakes
Commercial production
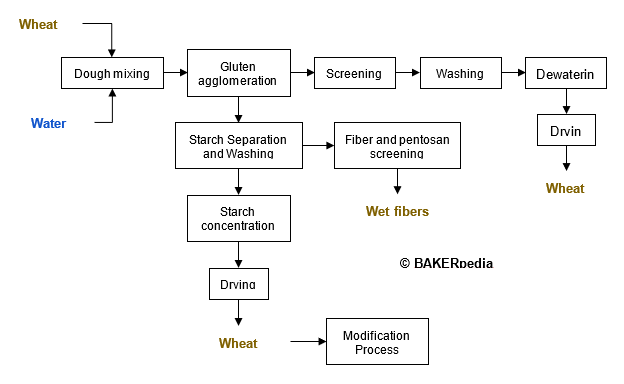
Modified wheat starch is produced from wheat flour according to the following block diagram:
Application
Chemical modification of wheat starch
Chemical modification of wheat starch involves the treatment of starch granules with small amounts of food-grade, chemical reagents. Like any other reaction process, agitation, temperature, pH, and reaction time become essential during the modification step.
- Depolymerization with acids (acid-thinned starch): Starch in granular form is soaked in acid to reduce gelatinization temperature, improve clarity of gel, reduce the tendency of gel to retrograde after cooling, reduce hot paste viscosity, and modify gel strength. Acid addition to starch causes a partial yet highly controlled hydrolysis which reduces polymer size of the polysaccharide, thus producing new reducing ends.2
- Stabilization: Stabilized starches are substituted with new side groups as replacers of hydroxyl groups. The substituents can be ethers (e.g., hydroxypropyl, hydroxymethyl) or esters (e.g., octenyl succinic anhydride, acetate, phosphate) and are added to granular starch. Stabilized starch properties are modulated by the exact nature of the substituting group: charged (polar) or uncharged, hydrophobic (non-polar), and whether it is ether- or ester-bonded.2
Once inserted into a starch granule, substituents prevent the same close packing of starch molecules. As a consequence, free water is better able to hydrate the granule, and so gelatinization temperature is decreased. During starch gel cooling, the substituents restrict the inter-chain associations of retrogradation. This confers improved freeze-thaw stability and a reduced tendency for syneresis. OSA-modified starch exhibits excellent encapsulating, fat replacer and emulsifying properties. OSA-modified starch is also a slowly digestible starch (SDS) with a low glycemic response.3
- Cross-linking: Cross-linking in granular starches is achieved by reacting –OH groups with di-functional reagents (e.g., sodium trimetaphosphate) that lead to cross-links such as di-starch phosphate esters and di-starch adipate (using adipic acid anhydride). Cross-linking reinforces the granule structure and makes starch granules more resistant to extreme temperatures (e.g. canning or retorting), high shear, and highly acidic food systems.4
Physical modification of wheat starch
- Thermal treatments
- Pre-gelatinization (precooked, instant starches)
- Granular cold-water-swelling starch
- Heat / moisture and mechanical shearing treatment (HMT)
- Annealing
- Dry starch heating
- Non-thermal treatments
- Intensive ball milling
- Ultrasonication
- High-pressure treatment
- Pulse electric field
Enzymatic modification of wheat starch
- Hydrolysis or enzymatic liquefaction (using different amylases)
- Dextrinization and saccharification for fructose and glucose syrup production
- Cyclodextrinization
Regulation
Modified starches are permitted in food products by the U.S. FDA. For chemically-modified starches, the quantity of any substance employed to affect such modification shall not exceed the amount reasonably required to accomplish the intended physical or technical effect, nor exceed any limitation prescribed. The Agency also lists the approved reagents which can be used in chemical-based modifications.
In the EU, 12 modified starches are approved for food use.5
References
- Schwartz, D., and Whistler, R.L. “History and Future of Starch.” Starch: Chemistry and Technology, 3rd edition, Academic Press, Elsevier Inc., 2009, pp. 1–10.
- Ross, A.S. “Starch in Foods.” Food Carbohydrate Chemistry, John Wiley & Sons, Inc., 2012, pp. 107–133.
- Luz Altuna, María Lidia Herrera, María Laura Foresti, Synthesis and characterization of octenyl succinic anhydride modified starches for food applications. A review of recent literature, Food Hydrocolloids, Volume 80, 2018, Pages 97-110.
- BeMiller, J.N. “Starches: Conversions, Modifications, and Uses.” Carbohydrate Chemistry for Food Scientists, 3rd edition, Woodhead Publishing, AACCI. Published by Elsevier Inc., 2019, pp. 191–220.
- U.S. Food and Drug Administration, CFR – Code of Federal Regulations Title 21, Part 172 — Food Additives Permitted For Direct Addition to Food For Human Consumption, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=172.892, Accessed 26 december 2020.
- EFSA 2017. http://www.efsa.europa.eu/en/efsajournal/pub/4911

