Sodium Benzoate
Also known as benzoate of soda
What is Sodium Benzoate?
Sodium benzoate is a common food preservative and a mold inhibitor. It is most effective in low acid foods and beverages and baked goods such as breads, cakes, pies, tortillas and many others.1
Benefits of sodium benzoate include its activity against:
- Molds
- Fungus
- Bacteria
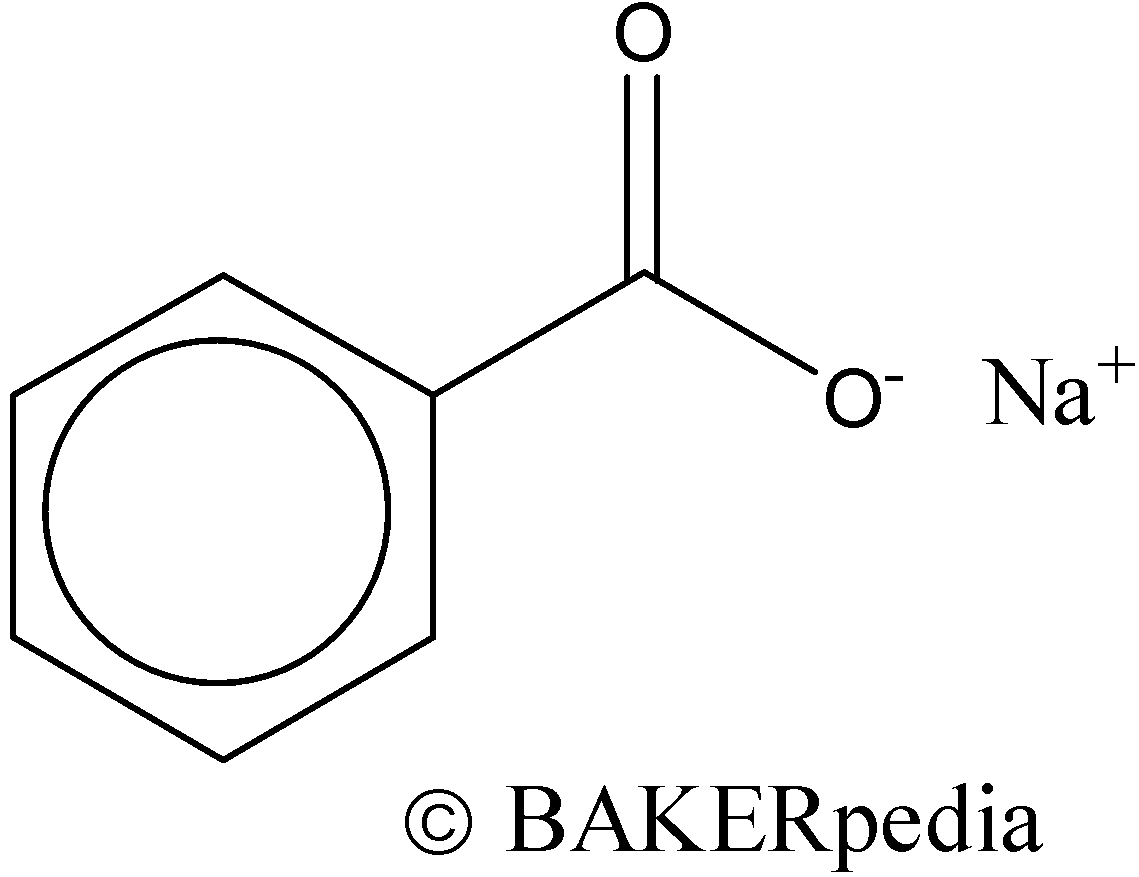
Chemical structure of sodium benzoate molecule:
Origin
The preservative activity of benzoic acid was described as early as 1875 by H. Fleck and was the first preservant permitted by the FDA. It is used in foods, cosmetics and drug formulations.
Function
Although benzoic acid is found in many plants, it is converted to the active sodium benzoate form to overcome its solubility challenges.
It is an effective preservative in baked products due to its activity against molds responsible for spoilage of most baked products. It is also used to control yeast, pathogenic and spore forming bacteria.2
In aqueous media and pH around 5.0, sodium benzoate transforms to benzoic acid which in the undissociated form can disrupt microorganisms’ cell wall. This retards their growth. At pH 4.0, 60% of the molecule is in the undissociated form compared to only 1.5% at pH 6.0.2
Nutrition
Despite the potential adverse effect of combining sodium benzoate and vitamin C and formation of benzene, the FDA states that levels of benzene are well below dangerous limits in properly formulated foods.3
Commercial production
Sodium benzoate is commercially produced using the following process1
- Neutralization: benzoic acid is mixed in a tank containing sodium hydroxide. The pH is controlled at 7.5-8.0 and a temperature of 95-98 °C (203-208 °F). The reaction is completed in 30-40 min
- Bleaching: removes undesirable colors
- Filtering: typically under pressure (0.3-0.4 Mpa) to obtain a clean solution
- Drying: it is oven-dried at 150-155 °C (302-311 °F)
- Packaging: particulates of 1.5-2.0 mm are packed in suitable containers
Application2,4,5
Similar to other preservatives, sodium benzoate can be mixed in the baked good formula or can be dusted onto the surface. Permitted usage levels of in food products are:
| Product | Recommended Level | Benefits | Drawbacks |
| Carbonated beverages | 0.02% | Prevents yeast spoilage | In presence of ascorbic acid and metal ions, may produce benzene in ppb concentration |
| Fruit juices | 0.05 – 0.1 % | Protects against mold and fermentation.
Usage with sulfur dioxide or other antioxidants increases antioxidation effect. |
Ineffective against oxidation and enzymatic spoilage |
| Pickles and sauerkraut | 0.1% | Highly effective at prevailing low pH | Risk of impairing the flavor |
| Mayonnaise | 0.05 – 0.1% | Imparts a stronger anti-bacterial effect when combined with potassium sorbate. | Risk of impairing product sensory properties |
| Baked goods | 0.1% | At low water activity and pH of 4.5 presents the optimal antimicrobial effect. | At high water activity (>0.8) only limited antimicrobial effect. |
| White layer cake | 0.1% | At pH value 6.4 presents the highest antimicrobial and antifungal effect in comparison with nisin and sulphite. | May slightly decrease cake volume. |
FDA regulations
Sodium benzoate is generally recognized as safe by the FDA, when used for its intended purpose.6
References
- “Sodium Benzoate.” National Center for Biotechnology Information. PubChem Compound Database. U.S. National Library of Medicine. https://pubchem.ncbi.nlm.nih.gov/compound/517055. Accessed 23 July 2020.
- Jay, J M., Loessner,M.J and Golden, D.A . Modern food microbiology.7 th ed., Springer Science & Business Media, 2005.
- Food and Drug Administration (FDA). US Department of Health and Human Services. “Chemical Contaminants – Questions and Answers on the Occurrence of Benzene in Soft Drinks and Other Beverages.” Questions and Answers on the Occurrence of Benzene in Soft Drinks and Other Beverages, Center for Food Safety and Applied Nutrition, https://www.fda.gov/food/chemicals/questions-and-answers-occurrence-benzene-soft-drinks-and-other-beverages, Accessed 24 July 2020.
- Guynot, M. E., Ramos, A.J., Sanchis, V. and Marín, S. “Study of benzoate, propionate, and sorbate salts as mould spoilage inhibitors on intermediate moisture bakery products of low pH (4.5–5.5).” International Journal of Food Microbiology 101.2 (2005): 161-168.
- Adeoye, B . “Comparative Evaluation of the Preservative Effect of Benzoate, Sulphite and Nisin on the Quality of White Layer Cake”. Greener Journal of Science, Engineering and Technological Research. 2. 048-052. 10.15580/GJSETR.2012.3.1212. (2012).
- Food and Drug Administration (FDA). US Department of Health and Human Services. CFR Code of Federal Regulations Title 21, Part 184 Direct Food Substances Affirmed As Generally Recognized As Safe, https://www.accessdata.fda.gov/scripts/cdrh/Cfdocs/cfCFR/CFRSearch.cfm?fr=184.1733, Accessed 23 July 2020.