
Butylated Hydroxytoulene
Also Known As BHT
What is BHT?
BHT is a phenolic antioxidant used in the preservation of a wide variety of products, including shelf-stable baked goods.
Origin
BHT is a synthetically aromatic organic compound derived from the reaction of 4-methoxyphenol and isobutylene, catalyzed by sulphuric acid. It was patented in 1947.1
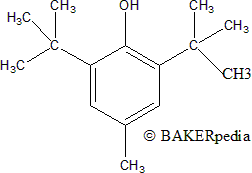
BHT chemical structure.
Function
BHT is used in foods that contain oil and fat. It functions as an antioxidant for preventing spoilage of fats and oils in baked goods by binding oxygen molecules. Oxygen molecules react preferentially with BHT rather than oxidizing the fats or oils. In addition, this action preserves food aroma, color, and flavor.
Application
Add directly to the shortening, as well as to baked goods containing fats, to prevent fat rancidity. BHT is not thermally stable. It evaporates at a temperature below 100oC, and evaporates/decomposes completely at 250oC.2 It has the least thermal resistance among synthetic antioxidants like PG (propyl gallate), BHA (butylated hydroxyanisole) and TBHQ (tertiary-butyl hydroquinone).2
It sometimes appears alone in a food, but often is used in combination with other chemicals that also have an antioxidant activity, including BHA, PG, TBHQ, citric acid, phosphoric acid, and ascorbic acid. Its effectiveness increases with the addition of synergists.
FDA regulation
BHT is GRAS. The FDA regulates the amounts used, or its combination with BHA, in foods in article 21CFR172.115 in the Code of Federal Regulations,3 as follows:
| Food | Limitations (total BHA and BHT) in parts per million |
|---|---|
| Dehydrated potato shreds | 50 |
| Dry breakfast cereals | 50 |
| Emulsion stabilizers for shortenings | 200 |
| Potato flakes | 50 |
| Potato granules | 10 |
| Sweet potato flakes | 50 |
The FDA also regulates in the same article that BHT must be labeled if it is used in food.
References
- Yehye, W. A., et al. “Understanding the Chemistry Behind the Antioxidant Activities of Butylated Hydroxytoluene (BHT): A review.” European Journal of Medicinal Chemistry, vol. 101, 2015, pp. 295–312., doi:10.1016/j.ejmech.2015.06.026.
- Santos, N. A., et al. “Commercial Antioxidants and Thermal Stability Evaluations.” Fuel, vol. 97, 2012, pp. 638–643., doi:10.1016/j.fuel.2012.01.074.
- U.S. Food & Drug Administration. “21CFR172.115 – Code of Federal Regulations Title 21.” Accessdata.fda.gov, 1 April 2017, www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm?fr=172.115. Accessed 5 September 2017.

